17th Annual Conference of the Ecological Society of Germany, Switzerland and Austria -
Section Plant Population Biology, May 2004 in Regensburg/Germany:
Population dynamics in a changing landscape - persistence, dispersal or adaption?
Spatial and temporal population dynamics and morphology of a violet hybrid in polluted pine forests of the Dübener Heide (Saxony-Anhalt)
Friederike Erlinghagen
UFZ Centre for Environmental Research Leipzig-Halle, Department of Community Ecology
Theodor-Lieser-Straße 4, D-06120 Halle, Germany
Correspondence: F. Erlinghagen, Wilhelm-Raabe-Weg 6, D-30938 Burgwedel, Germany
E-Mail: infoogzpLw@friederike-erlinghagen.de
Abstract
The investigations were focused on the demography and morphology of the clonal perennial hybrid Viola riviniana RCHB. x Viola reichenbachiana JORDAN ex BOR. (V. x intermedia RCHB., V. x bavarica SCHRANK) in central Germany . In particular, the aims of the study were to investigate quantitatively the importance of sexual and clonal propagation with regard to the local dynamics of the hybrid and to evaluate the consequences of propagation patterns and morphological characters on adaptation to the changed environment of polluted pine forests.
The demographic study was carried out in six violet populations of different ages under different habitat conditions: young spreading populations of the hybrid in polluted pine forest sites and old established populations of Viola riviniana in an isolated deciduous forest site were compared. A number of differences were identified with regard to seedling recruitment between the pine forest sites and the deciduous forest sites. In both of the latter, evidence was found for repeated seedling recruitment in addition to clonal growth in Viola populations. The results indicate that clonal propagation is accompanied by high ramet mortality (high annual turnover). In pine forest populations seedling germination is partly a result of seed limitation, whereby singly-accumulated and early germination leads to density-dependent mortality. Here, the seed weight seems to play an important role for survival and competitiveness. The weaker correlation between seedling recruitment and fruit production as well as positive density-dependency indicate that the variation in microsite quality is more important for older populations in deciduous forest sites. The established and isolated populations in deciduous forest sites produce seeds that are significantly lighter than those of pine forest populations. As a consequence, their resources are fewer, which makes their germination and establishment far more dependent on varying microsite quality. As a consequence of abortion and “genetic load” it may additionally be concluded that seeds are more adapted in these microsites.
Despite genetic and demographic differentiation, the investigations indicate a number of mutual characteristics between all violet populations: resource allocation capability and the resulting high costs of reproduction lead to a short ramet life span, thus raising the question of an optimum life-history strategy. Both the present demographic studies as well as the molecular-genetic investigations of Neuffer et al. 1999 lead to the hypothesis that introgressive hybridization results in novel genotypes adapted to the changed environment of polluted pine forests. It will be assumed that the Viola populations of the pine forests have “colonized their respective sites during the past few centuries” (in citing Neuffer et al. 1999). But the results raises the question of the introgressive hybridization of Viola species described to aerial pollutions occurred not until the last decennia. Viola riviniana and Viola reichenbachiana coexist in the isolated deciduous forest sites within the pine forests of the Dübener Heide, so that from this hybridization and invasion is possible.
Keywords: Clonal growth, environmental pollution, introgressive hybridization, seedling recruitment, trade-off, Viola riviniana
Introduction
Clonal plants are capable of producing genetically identical but potentially independent ramets which form a genet or clone (Klimes et al. 1997). An important difference compared to nonclonal organisms is the differentiation between physiological and genetic individuals. In Central Europe clonal growth occurs in 2/3 of all plant species (Klimes et al. 1997).
The importance of seedling recruitment in populations of clonal plant species is the subject of numerous investigations (e.g. Eriksson 1989, Auge 1997). The colonization of or establishment at a new site is related to the dispersion of seeds in most cases. Two possibilities exist for the further development of the population: 1. Further seedling recruitment does not take place after the initial colonization (“initial seedling recruitment“=ISR), and 2. Repeated seedling recruitment (“repeated seedling recruitment“=RSR) (Eriksson 1989). The extent to which a species repeatedly reproduces at a new habitat has consequences for selection, e.g. for the genetic diversity within a population, and hence for the evolution of the life cycle (Bierzychudek 1982).
The present investigation deals with the introgressive hybrid populations between the clonal tetraploid species Viola riviniana RCHB. and the clonal diploid species Viola reichenbachiana JORDAN ex BOR. (wood violet), which become established and have spread in the polluted pine forests of the Dübener Heide. The spreading of the Viola populations thereby correlates with the effects of pollution on habitat conditions (Neuffer et al. 1999).
Hybridization between plant species occurs frequently. Besides primary evolution factors such as mutation, genetic recombination and selection, secondary factors such as fluctuations in the population size, isolation mechanisms, annidation (niche colonization, i.e. the discovery of a non-competitive niche in the biotope) and hybridization between species are considered to be important mechanisms influencing speciation (synthetic theory). Hybridization mostly results in mixed forms, and many arguments suggest that these hybrids mainly exhibit lower fitness than their parental, pure species (Stace 1975, Arnold 1997) and are frequently restricted to ecotones or disturbed habitats. Contrary evidence, however, also exists (e.g. Rieseberg 1995). For example, forms with new characteristics occasionally occur, and hybridization has in fact led to invasive genotypes (Daehler & Strong 1997) or new species (Abbott 1992).
Within the scope of the present work an attempt is made, with the aid of population ecological methods under consideration of the life cycle, to investigate the ecological niche colonization of Viola riviniana x Viola reichenbachiana in the changed abiotic and biotic environmental situation in pine forests affected by atmospheric pollution. For the purpose of comparison, populations of the pure species Viola riviniana in a deciduous hardwood forest (cf. molecular-genetic investigations by Neuffer et al. 1999) were also considered in the study. Particular attention is given to the following questions:
- Are there differences in the spatial and temporal development of the Viola populations between the polluted pine forest sites and the deciduous forest sites?
- Are there differences in the growth and reproductive behaviour of the ramets and what kind of influence do these have on the spatial-temporal dynamics of the Viola populations?
- Is it possible to recognize adaptation mechanisms leading to an establishment of populations in the polluted pine forest sites?
Materials and methods
The biology of Viola riviniana
Viola riviniana RCHB. is a frequently occurring rosette plant, widespread in forests and meadows (Grime et al. 1988). It exhibits two modes of vegetative propagation: 1) by branching rhizomes, which rarely occurs (Meusel & Kästner 1974) and 2) by the production of adventitious root buds. Moreover, new rosettes blossom from the axilla of the old rosettes of the rhizomes lying on the ground (Meusel & Kästner 1974). Viola riviniana is capable of cross-pollination as well as self-pollination. The relatively long-spurred chasmogamous flowers appear in spring and are pollinated by long-proboscis Apoidea (e.g. various bumblebees). The small, almost unnoticeable cleistogamous flowers succeed the chasmogamous spring flowers relatively late in the year in June-September (Grime et al. 1988, Beattie 1969, Berg & Redbo-Torstensson 1999, season-related phenology after Hess 1983). The dispersion of the seeds is diplochoric (Beattie & Lyons 1975, Grime et al. 1988). Firstly, the seeds are dispersed by a catapult mechanism, whereby dispersal only occurs over small distances of 4-6 m (Müller-Schneider 1986). Secondly, the seeds possess a typical eleiosome and are dispersed by ants (myrmecochory). The majority of seeds originates from cleistogamous flowers (Beattie 1969), whereas chasmogamously produced seeds have a higher germination rate, resulting in a larger number of seedlings (Berg & Redbo-Torstensson 1999). Compared to Viola riviniana the closely-related Viola reichenbachiana has a narrower niche and inhabits sites that are less acidic and richer in nutrients (Valentine 1949, Meusel et al. 1978, Oberdorfer 1990, Rothmaler 1997).
Study sites
The investigations were carried out at two forest sites in Saxony and Saxony-Anhalt (Central Germany), representing two different types of Viola riviniana populations and plant communities. The study sites S1-S4 lie within the pine forests (Pinus sylvestris plantations) of the Dübener Heide north of Leipzig. The distance between the study sites varies from 5 to 14 km.
The input of calcareous particulate depositions since the beginning of the 20th century has resulted in an increase in the pH-values of the soil (Neumeister et al. 1991). Sulphur dioxide emissions have resulted in damages to the tree crowns. Improved light infiltration as well as increased base saturation and improved nutrient supply due to urea fertilization of the formerly nutrient-poor and acidic, sandy soils of this area have led to a change in the abiotic and biotic environmental conditions in the pine forests. The changed plant communities are characterized by the invasion of alien plant species, such as Mahonia aquifolium (Auge 1997, Auge & Brandl 1997), as well as by more demanding deciduous forest species, which has consequently affected the associated food web (Brändle et al. 2001). Viola riviniana, a typical deciduous forest species is characterized by the introgression of Viola reichenbachiana genes in the pine forests of the Dübener Heide (Neuffer et al. 1999). The Viola populations most likely became established in the second half of the 20th century and thus represent a relatively young population (Neuffer et al. 1999). Due to the fact that changes in vegetation still persist today, these areas may be considered to be highly dynamic systems.
The two study sites P1 and P2 are located in a deciduous forest on “Petersberg” within the nature reserve “Bergholz” north of the town of Halle. Quercus petraea (chestnut oak), Carpinus betulus (hornbeam) and Tilia cordata (European lime) dominate in the tree layer; the plant community may be classified as a lime-rich Galio-Carpinetum (chestnut oak – hornbeam forest) (Große 1985). The association grows on hilly cultivated land on fairly dry soils, whereby the “Bergholz” represents a remnant of a formerly more widespread association. During the 12th century, the woodland between Petersberg and Gutenberg was felled, apart from a few residual forests such as e.g. the “Bergholz” (Große 1985). The deciduous forests represent an old, isolated but stable system with well-established Viola riviniana populations. The two study sites are characterized by loess soil with low pH values in its uppermost layers (Erlinghagen 1998). Sites P1 and P2 are 0.25 km away from each other and located 32-45 km from the study sites S1-S4.
All of the selected pine forests are old stocks (> 40 years). The degree of coverage of the herb layer was about 80% at all study sites in July 1995; with the exception of the study site S3, large amounts of dead biomass accumulate in summer. Only study site S4 exhibits a shrub layer interspersed with gaps.
Study site S1: herb layer with Calamagrostis epigejos dominance. Within the study period high invasion of Rubus idaeus (raspberry) and Rubus plicatus agg. (blackberry) occurred. The moss layer is relatively open.
Study site S2: herb layer with Calamagrostis epigejos, Rubus idaeus and Rubus plicatus, with an open moss layer.
Owing to the presence of Brachypodium sylvaticum and Fragaria vesca (wild strawberry) the two study sites can be classified as Brachypodium-Fragaria pine forests (cf. Wiegand & Amarell 1995).
Study site S3: apart from Brachypodium sylvaticum and Rubus species, the site is characterized by Vaccinium myrtillus (bilberry) and bryophytes; the moss layer increased considerably during the investigation period and it partly smothered other vegetation.
Study site S4: apart from Calamagrostis epigejos, Vaccinium myrtillus, Rubus idaeus and Rubus plicatus agg. the study site is characterized by Pteridium aquilinum and Rubus caesius. The presence of these species possibly suggests lower-lying, small-area clay bands or high eutrophication. The moss layer is well developed.
Study sites P1 and P2 in the deciduous forest of the nature reserve “Bergholz”: lime-rich Galio-Carpinetum with a rich developed herb layer. Anemone nemorosa (wood anemone) predominates in spring, whereas in summer, Galium odoratum (woodruff), Circaea lutetiana and Impatiens parviflora attain high cover degrees. The total cover of the herb layer in summer is about 80 %. Dead biomass accumulates in the spring, but almost completely degrades during summer. The moss layer is only poorly developed.
Demography
In order to estimate the density of ramets, fruit production and seedling recruitment, permanent plots of 2x2 m were established. Three of the study sites were set up in spring 1994 (S1,S2 and S3) and three in spring 1995 (S4, P1 and P2). Depending on the density of the Viola populations, the number of permanent plots varied between four and nine. A reduction in the number of specimens for very high ramet density appeared to be more advantageous compared to a reduction in the plot size, as it was also intended to investigate the spatial dynamics of the ramets and seedlings parallel to their temporal dynamics. The spatial arrangement of the plots within each study site followed the stratified unaligned sampling method (cf. Greig-Smith 1983), whereby 1-2 plots were established in each 10x10m grid. As the spatial distribution is dependent on the previously considered scale, each plot was subdivided uniformly into 100 0.2x0.2m subplots. All Viola ramets and seedlings within the plots and subplots were marked and temporally and spatially monitored about every 2-3 weeks during the vegetation period up to autumn 1996. The number of fruits within the plots was recorded each year.
Due to the fact that the rhizome of Viola grows as a monopodium over many growth periods and does not exhibit persistent branching (Meusel & Kästner 1974), the visible part of the perennial axil above the ground was defined as the counting unit in the present study and referred to in the following as a ramet. By this means it was possible to quantify vegetative propagation on the basis of the rhizome and root sucker. Rosettes flowering out of the leaf axils of the old rosettes were thereby not evaluated as ramets.
Morphology
In order to estimate the generative reproduction of the clonal species the number of seeds per fruit and the weight of each seed were determined. For this purpose, fruits were collected from the study sites in 1996. Only one fruit was removed from each ramet; in total, fruits were collected from 70-100 ramets per study site. The seeds were dried at 60 °C over 5 hours after which the dry weight was determined with the aid of an analytical balance.
The program GLIM 4 was used for the statistical evaluation of density dependence. Variance analyses (ANOVA), LSD tests, correlation analyses (Pearson) and simple regression analyses were carried out with the aid of the program WINSTAT. The dispersion of the ramets and seedlings was computed on the basis of the variance/mean ratio (dispersion index) over the plots and subplots. Accordingly, a value of s²/x < 1 indicates a uniform distribution, s²/x = 1 Þ a random distribution, and s²/x > 1 Þ clumping. In order to assess whether the dispersion of the ramets and seedlings deviated significantly from the Poisson distribution (i.e. from the random distribution) the Chi² test was applied and the values were finally transformed (z-transformation, cf. Krebs 1989).
3 Results
Demography
Seedlings
The seedling density differed considerably between the populations and was generally higher in the pine forest than in the deciduous forest (p<0.01 1995, p<0.001 1996) (Table 1).
Table 1
Density of ramets, fruits and seedlings in 4m² plots within 6 Viola populations (mean value ± SD). The study sites S1-S4 are located in pine forests whereas P1 and P2 are study sites located in deciduous forests
| No. of ramets/plot | No. of fruits/plot | No. of seedlings/plot | |||||
|---|---|---|---|---|---|---|---|
| - | 1995 | 1996 | 1994 | 1995 | 1995 | 1996 | |
| Site | No. of plots | ||||||
| S1 | 4 | 265±130 | 218±113 | 88±40 | 114±33 | 195±93 | 1018±579 |
| S2 | 7 | 50±29 | 54±20 | 12±12 | 9±10 | 58±55 | 53±38 |
| S3 | 5 | 73±40 | 38±16 | 65±33 | 2±2 | 218±163 | 19±12 |
| S4 | 8 | 25±13 | 30±21 | 13±8 | 22±11 | 43±31 | |
| P1 | 9 | 31±35 | 23±21 | 8±10 | 22±19 | 19±15 | |
| P2 | 6 | 57±40 | 39±22 | 14±10 | 12±8 | 10±7 | |
| All sites | 39 | 83±90 | 67±75 | 55±28 | 27±43 | 88±94 | 194±404 |
The annual fruit production between the populations also varied, whereby seedling density correlated with fruit production in the previous year within the 2x2m plots. In 1994 the pine forest populations S1 and S3 produced most fruits per plot, differing significantly from the pine forest population S2 (p< 0.001). Whereas the higher number of fruits in population S1 resulted from the higher number of ramets, the ramets of population S3 produced more fruits per ramet, differing significantly from populations S1 and S2 (p<0.001). In the subsequent year both populations exhibited the highest seedling density (p<0.001). In the pine forest population S3 the production of generative organs was almost completely absent in 1995, whereas population S1 again exhibited the highest seedling density (p<0.001) (Erlinghagen 1998, Erlinghagen in: Auge et al. 2001). This result suggests that i) most seeds are dispersed over only a short distance and ii) the recruitment of new seedlings is seed-limited (Eriksson 1992, Turnbull et al. 2000). The correlation between fruit production (1995) and seedling emergence (1996) was less pronounced in the deciduous forest (r=0.49, p<0.05, n=15) than in the pine forests (r=0.83, p<0.001, n=24); a difference which is marginally significant (p=0.08).
The seeds were found to germinate within a relatively short period of time, and all seedlings had germinated by late spring in both the pine forest and deciduous forest populations. The survival rate of the seedlings in the deciduous forest populations was found to be significantly higher than in the pine forest populations (Table 2). A comparison among pine forest populations revealed a higher survival rate of seedlings in populations S2 and S4 after the first few months. Whereas the survival rate of seedlings in population S2 no longer differed from the other populations after one year, population S4 was found to have a more than threefold higher survival rate of 26%.
Table 2
Seedling survival rates in 1994 and 1995 (No. of seedlings in April, August after the first and second year). All values refer to the number of seedlings in spring (=100%). Note: due to the fact that the seedling densities were very high in several cases, the size of the individual samples was partly reduced for computing survival rates. The percentages of survived seedlings are thus referred to the overall number of seedlings counted per site, i.e. it was not possible to carry out a significance test
| Seedlings 1994 | Seedlings 1995 | |||||
|---|---|---|---|---|---|---|
| - | Aug | Year 1 | Year 2 | Aug | Year 1 | |
| Type | Population | |||||
| Pine forest | S1 | 32% | 5.9% | 2% | 13% | 5.1% |
| S2 | 65% | 13.2% | 1.3% | 20% | 6.5% | |
| S3 | 34% | 2% | 1.3% | 15% | 8% | |
| S4 | - | - | - | ? | 26.5% | |
| Deciduous forest | P1 | - | - | - | 55% | 22% |
| P2 | - | - | - | 37% | 24.6% | |
| Mean value | - | - | - | 46% | 23.3% | |
At the scale of the plots (dispersion index 7.3-42) as well as at the scale of the subplots (dispersion index 1.2-6.2) the seedling emergence was clumped. The effect of seedling density on the seedling survival rate was contingent on habitat site at the scale of the subplots. In the pine forests the seedling survival decreased with increasing density in spring, which may be explained by increased intraspecific competition. In contrast to the latter, the survival rate in the deciduous forest increased with increasing seedling density. The populations within a habitat type do not differ (interaction of habitat type density after accounting for population and plot effects in the logistic regression model: log likelihood ratio = 10.46, 1 d.f., p<0.005). This result suggests that seedlings in the deciduous forest are primarily concentrated to the most favourable microhabitats. Accordingly, safe sites play an important role in seed germination and seedling growth. Up to the end of their first growth period the average survival rate was 0.11 ± 0.10 and 0.23 ± 0.02 (mean ± SD) in the pine forests and the deciduous forests, respectively (Erlinghagen 1998, Erlinghagen in: Auge et al. 2001).
Ramets
With regard to ramet density, no significant differences existed between pine forest and deciduous forest populations. Although ramets in the pine forests partly exhibit very high densities (S1, cf. Table1), the variance between the populations in the pine forests was nevertheless larger than the variance between the two habitat types. The ramet density remained relatively constant in all investigated Viola populations in spring over the entire study period: only the pine forest population S3 exhibited a slight decrease. The number of ramets counted in spring of the respective study year thereby represented the sum of ramets surviving the previous winter and the clonally-produced ramets in spring. Within the pine forest populations, population S1 was characterized by a very high ramet density in spring and differed significantly from the other pine forest populations (p<0.05 1994, p<0.001 1995 and 1996). Up to the end of the growth period in 1995 the ramet birth rate (clonal growth due to rhizome branching and adventitious root buds) was 0.083 ± 0.048 (mean ± SD between all populations) (Erlinghagen in: Auge et al. 2001), whereby the pine forest population S1 produced several generations up to autumn in the first study year; a phenomenon that was not observed in other populations (Erlinghagen 1995). In the subsequent year the ramet density continued to increase up to June, and compared with the other populations, attained a more than two-fold higher vegetative propagation rate of 15% (Table 3). The mean ramet survival probability of 0.40 ± 0.19 up to the end of 1995 was very low (Erlinghagen in: Auge et al. 2001). Despite an unusually high growth rate up to the time of fruit ripening in June, the pine forest population S3 exhibited the highest “death rate” compared to the other populations, with ramets hardly producing any flowers or fruit. About 75 % of the ramets had “died” up to August within this population as well as in the pine forest population S1, whereas in the pine forest population S2 and the deciduous forest population, the “death rate” was approx. 40% or 50% respectively.
Table 3
Vegetative reproduction rate and “death rate” of ramets
| Vegetative | "death rate" up to | ||||
|---|---|---|---|---|---|
| Habitat type | Population | reproductive rate 1995 | 06.95 | 08.95 | |
| Pine forest | S1 | 15.0% | 0.75% | 72.5% | |
| S2 | 5.6% | 13.2% | 49.5% | ||
| S3 | 7.5% | 23.1% | 78.0% | ||
| Deciduous forest | P1 | 3.8% | 10.9% | 37.4% | |
“Death” in this context is understood to be the loss of ramet biomass above the ground, as it was not possible to determine the extent to which the apical meristem of a rhizome had also died up to this point in time. Due to the fact that several of these ramets again produced leaves at a later date (1996), the computed survival probability represents a slight underestimation.
In overall terms it may be concluded from the observations that these birth and “death” rates indicate a high turnover of ramets within a single year. A short ramet life span is also substantiated by the fact that the seasonally-related phenology of flowers was only slight, cleistogamous flowers emerged relatively early in the year, and no additional flowers developed after June.
At the scale of 2x2m the ramets exhibit high aggregation within the populations (variance to mean ratio of 7.2-64), but only slight aggregation on the subplot scale of 0.2x0.2m (dispersion index 1.2-2.0). At the scale of 2x2m the deciduous forest populations with the lowest ramet density partly exhibited high dispersion values (strongly aggregated). A comparison between the pine forest and deciduous forest populations at the scale of the subplots showed only a weak relationship between ramet density and z-values in the pine forest (r=0.29, p<0.05 Spearman rank correlation), whereas a pronounced relationship existed between the values in the deciduous forest (r=0.64, p<0.001). If the differences are considered differentially according to different periods of time (spring and autumn 1994, June 1995), z-values and the number of ramets were found to be independent of each other within the pine forest populations. With a doubling of the ramet density the dispersion in population S1 during the course of 1994 was found to either remain constant or decrease significantly. This results thus seems to indicate that in the pine forest population S1 the vegetative propagation rhythm combined with a short ramet life span plays an important role in the rapid spatial propagation of phalanx strategists. In the deciduous forest populations, on the other hand, the spread of ramets appears to reflect the variance of microhabitat quality, as already established for seedlings.
Morphological differences
Ramets
Multivariate analyses of the morphological data (petal size and colour, the length, width, surface and colour of the petal spur, the length of sepal appendages and stipular fringes) indicate intermediate characteristics, characteristics of the putative parental taxa as well as a number of extreme characteristics (e.g. length of the petal spur) (Neuffer et al. 1999). Pronounced morphological differences were exhibited especially by the ramets of the pine forest population S1 regarding their size (height and leaf size). Unusually small ramets frequently grew in the root zone of pine trees; the size of the ramets also varied significantly in the Calamagrostis population. It is thus probable that the high size variability of the ramets in the pine forests is the result of a microhabitat differentiation or a different rate of reproduction rather than age differences.
Following high generative reproduction in 1994 the ramets of the pine forest population S 3 exhibited unusually high growth in the subsequent year (leaf size, ramet height, stem girth), whereby the generative phase was almost completely absent. Due to the fact that a very high bryophyte growth rate occurred at this habitat in particular, the observations suggest that the compensation of an increased competitive capability serves to guarantee ramet survival. A large proportion of the ramets nevertheless died in spring.
Weight and number of seeds
In many cases the number of seeds influences the maternal fitness directly, whereas the seed weight frequently affects the offspring (Haig & Westoby 1988). Due to the fact that seedling recruitment in the pine forest populations is seed-limited and that density-dependent mortality of the seedlings could be verified, it is probable that seed quality plays an important role in seedling survival. The size of a seedling and hence its chances of survival and competitive capability is dependent on seed weight in many cases. Moreover, seed weight is important for the systematic classification of the investigated Viola populations.
With a mean seed mass of 1.1mg the ramets of the pine forest populations produced significantly heavier seeds than the ramets of the deciduous forest populations with average values of 0.89 mg (p<0.05, Table 4). The ramets of population S2 had the heaviest seeds, with an average 1.41mg (p<0.001), whereas population S1 had the lightest seeds with 0.91mg (Table 4). The deciduous forest populations produced seeds as light as those of the pine forest population S1, averaging 0.94 and 0.85mg for the populations P1 and P2, respectively. Both differed significantly from the pine forest populations S2 and S4.
Table 4
Mean seed weight and mean no. of seeds/fruits ± SD in June 1996. ANOVA (LSD Test: *: p<0.05, **: p<0.01, ***: p<0.001)
| Seed weight (mg) | No seeds/fruit | ||
|---|---|---|---|
| Pine forest | 1.1±0.49* | 14.2±5.1 | |
| Deciduous forest | 0.89±0.29 | 17.9±6.9*** | |
| F-Value | 360.4 | 28.5 | |
| Pop. | S1 | 0.91±0.43 | 13.4±22.8 |
| S2 | 1.41±0.4*** | 12.3±3.8 | |
| S4 | 1.14±0.5*** | 16.4±5.3*** | |
| F-Value | 325.5 | 16.1 | |
| Pop. | P1 | 0.94±0.26 | 16.7±6.9 |
| P2 | 0.85±0.3 | 19.1±6.7 | |
| F-Value | - | - | |
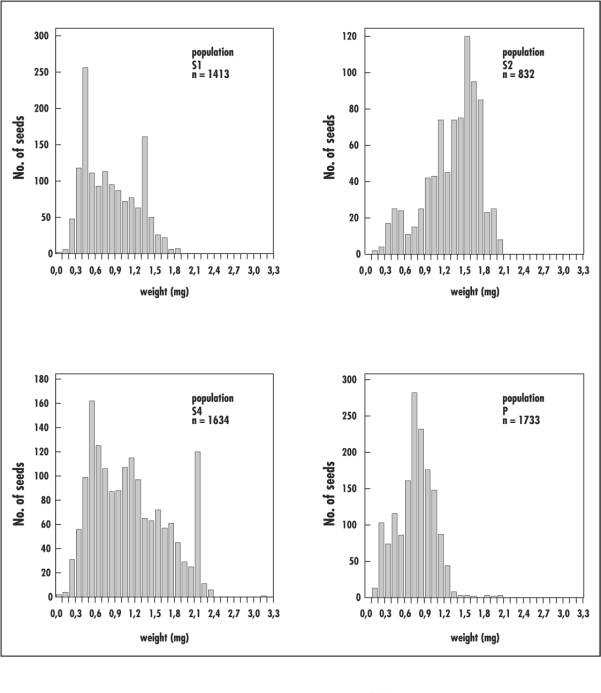
All investigated Viola populations produced some seeds with very low mass of about 0.3mg; the heaviest seeds, weighing 3.2mg, were found in pine forest population S4. Depending on their weight, the seeds were also found to differ in colour. Light seeds were white, medium-heavy seed mostly light brown, and heavier seeds dark brown. The weights of individual seeds within one fruit were relatively constant, with differences seldom exceeding 0.2mg. The differences in mean seed weight between individual fruits were partly very small; on the other hand, fruits with heavy seeds were also found alongside fruits with light seeds on a single ramet.
The frequency distribution of seed weights showed that the pine forest populations S2 and S4 tended to produce heavier seeds, whereas in the pine forest population S1 and the deciduous forest populations (P1 and P2 lumped together) intermediate characteristics are often present (Figure 1). Similar seed weights and seed frequency distributions in populations of the different habitat types S1 and P suggest that seed weight is not governed by habitat factors alone.
At the deciduous forest sites the ramets produced significantly more seeds/fruit (17.9 on average) than the ramets at the pine forest sites (14.2 on average) (p<0.001) (Table 4). A comparison among the pine forest populations showed that population S4 produced most seeds/fruit (p<0.001), differing only slightly from the deciduous forest populations.
Summarizing, the ramets of the deciduous forest populations produce significantly more and lighter seeds than the ramets of the pine forest populations. A comparison between the pine forest and deciduous forest sites indicates, however, that seed weight has no bearing on the number of seeds produced in a single fruit. Under differentiated consideration of the pine forest populations it was, however, possible to verify a weakly negative but highly significant relationship between mean seed weight and the mean number of seeds per fruit for population S2 with the highest mean seed weight (r = -0.34, p<0.001).
Discussion
Repeated seedling recruitment in populations of clonal plant species is generally more the exception than the rule. Repeated seedling recruitment was evident in 40 % of the 68 clonal plant species compiled by Eriksson (1989), whereby a negative correlation exists between the population growth rate and vegetative/sexual reproduction (Silvertown et al. 1993). Demographic studies of several clonal species have shown that repeated seedling recruitment is as important as vegetative propagation for maintaining the populations of these species (Bierzychudek 1982, Inghe & Tamm 1985). In this context initial seedling recruitment and repeated seedling recruitment should be considered as two extremes within a continuum, i.e. as two, clearly distinct recruitment patterns in clonal plant species (Eriksson 1993). The short-period study presented here shows that repeated seedling recruitment occurs in conjunction with clonal propagation in all investigated Viola populations. Repeated seedling recruitment has also been verified for the deciduous forest species Viola blanda, V. pallens (Newell et al. 1981) as well as Viola sororia (Solbrig 1981) in the USA. In agreement with demographic results, and independent of habitat type and population age, molecular investigations with the aid of random amplified polymorphic DNA analyses (RAPD) revealed high genotypic diversity within local Viola populations (Auge et al. 2001). High genotypic diversity is in fact retained, regardless of whether the mortality rate of seedlings at the pine forest sites are positively density-dependent. It is clear that none of the genotypes are capable of producing sufficient clonal offspring to influence their dominance (Auge et al. 2001). Simulation models have shown that even a low rate of repeated seedling recruitment is sufficient to prevent the loss of genotypic diversity within clonal plant populations (Watkinson & Powell 1993).
Within the pine forest and deciduous forest populations it was possible to verify a number of differences regarding seedling recruitment: the weaker correlation between the number of seedlings and fruit production as well as the positive density-dependent survival probability of the seedlings suggest that the variation in microhabitat quality is more important for the established deciduous forest populations than for the young pine forest populations (Erlinghagen 1998, Erlinghagen in: Auge et al. 2001). The majority of seedlings emerged in spring in all of the investigated Viola populations within a relatively short period of time, whereby a large proportion died up to the summer. A negative exponential mortality of seedlings could also be verified for Viola blanda and Viola sororia (Solbrig 1981). The single, aggregated and early germination within the pine forest populations results in density-dependent mortality. The advantage of this germination process is the early life commencement of the seedlings. In overall terms the pine forest populations produce significantly heavier seeds than the deciduous forest populations, whereby populations S2 and S4 exhibit the heaviest seeds and, at the same time, the highest seedling survival rate during the first year. There is evidence to suggest that the seed weight within a pine forest population plays a role in the chances of survival and the competitive capability of the seedlings. Studies of Viola sororia confirm that seedling survival is a function of size (Newell et al. 1981).
Generally speaking, a distinction is made between two colonization strategies of clonal-growth plants (Lovett-Doust 1981): in the case of the guerilla type, clonal growth results in a spatial distribution of ramets. With their long rhizomes and stolons, guerilla strategists grow opportunistically and avoid the competition posed by other species. Contacts are thereby mainly intraspecific and relate either to the same species or treat the same species as a foreign species (intra- and interspecific) (Urbanska 1992). In the case of the phalanx type clonal growth leads to a compactly developed plant due to concentration of the shoot system; by this means, phalanx species generally have a high competitive capability. Typical representatives are e.g. tufted grasses. In the case of this strategy type neighbouring species are mostly confined to the interspecific level. Accordingly, neighbouring individuals mainly belong to the same clone, and further intra- and interspecific contact normally only occurs on the boundary of a population. Both strategy types are extreme forms of a continuum, between which transitions exist. Viola riviniana is rather classified as belonging to the phalanx type (Grime et al. 1988). The ramets of all investigated populations exhibit a similar strongly aggregated spatial pattern on both scales, whereby the z-values in the pine forest populations were independent of the ramet density, and aggregation in the forest pine population S1 in fact reduced with a two-fold increase in the ramet density. Due to the fact that several generations were produced in population S1 during the year of observation (1994) the results suggest that a rapid spatial propagation occurred within this population especially due to clonal growth and, in particular, due to the development of rhizome and root suckers. The spatial distribution of the ramets is thereby especially governed by the species-specific propagation mode, as already established for seedling recruitment. This takes place by way of an opportunistic strategy, as characteristic of guerilla strategists. Calamagrostis epigejos is considered to have high competitive capability. Ramets of the Viola hybrid spreads in gaps, and as long as gaps occur in sufficient number, a weakly competitive (Grime et al. 1988) and a strongly competitive species may coexist, whereby they may probably be displaced just as quickly due to succession.
Life histories represent ecological strategies which develop on the basis of the selection pressure of different habitats and guarantee optimum fitness of a species under given habitat conditions. Reproduction and mortality are the two essential fitness components. The quantitative importance of reproduction and mortality for the fitness of a species varies according to life history. The life histories thereby represent different possibilities of dealing with limited resources, which must be distributed among competitive plant activities and structures (“principle of allocation” Cody 1966, Harper & Ogden 1970). This means that e.g. growth and reproduction compete for resources and life span, i.e. reproduction basically occurs at the cost of vegetative growth. A fundamental “trade-off” thus exists between these two processes, which eventually leads to a “trade-off” between the survival probability and reproduction of individuals (Grime 1979, Scheiner 1993). Accordingly, the decline of vegetative growth due to reproduction leads to reduced competitive capability and hence, indirectly, to an increased mortality risk. Reproduction may, however, also be the direct cause of death of a plant, as is basically the case in semelpare reproduction. Negative correlations between life cycle characteristics (“trade-off”) may also be assessed as “reproduction costs” (Luken 1987) or reproduction costs may be the result of a “trade-off” (Stearns 1976).
In all of the investigated Viola populations it was possible to verify a high annual turnover of ramets, which may be explained by a high production of clonal offspring and a considerably high ramet mortality. The size of the population over the study period thereby remained relatively constant; an exception to this was the pine forest population S3. A very similar behaviour was exhibited by the populations e.g. of Viola blanda (Newell et al. 1981) and Hieracium pilosella (Bistop et al. 1978). In the case of these species it was possible to verify that a synchronous birth and death rate led to a constant population size (cf. Cooke 1985: in Jackson et al.). Evidence for a density-dependent regulation of mortality could not be verified, however. The short life span of the ramets suggests high reproduction costs. The present investigations thus indicate trade-off situations between vegetative propagation and generative reproduction (pine forest population S3), between clonal propagation and survival probability (production of several generations in the pine forest population S1) as well as between the number and weight of seeds (pine forest population S2). Heavier seeds were thereby preferably produced in the pine forest populations S2 and S4; seeds with a weight of 3.2mg are not known for Viola reichenbachiana, which develops heavier seeds than Viola riviniana. In addition to the extreme morphological characteristics determined by Neuffer et al. 1999, the heavy seed weight may be assessed as an additional extreme characteristic, and as an indication of a possible evolutionary “trade-off”. If the production of heavier seeds at the cost of the number of seeds is evaluated as an evolutionary “trade-off” and not as a pure introgression of characteristics, this may indicate an adaptation to the habitat, which raises the question of genealogical differentiation. The reproductive effort generally appears to be larger for species or strains in disturbed habitats than for species which essentially propagate in relatively stable habitats. This is indicated by a comparison of the reproductive effort of different Viola species (Fitter & Setters 1988), Solidago species (Abrahamson & Gagdil 1973), as well as Galium aparine populations (Auge 1991) and Taraxacum officinale (dandelion) populations in habitats with varying degrees of disturbance (Solbrig & Simpson 1974).
For the pure species Viola riviniana Valentine (1941) found the seed weight to vary between 1.0 and 2.1 mg, whereby forest populations with average seed weights of 1.4-2.1mg produce heavier seeds than populations colonizing open habitats (average seed weight 1.0-1.3mg). It was only possible to verify lower seed weights for the hybrids Viola riviniana RCHB. x Viola reichenbachiana JORDAN ex BOR. (Viola bavarica SCHRANK) with Viola riviniana as the female parent. For example, the hybrid x riviniana and vice versa produces seeds weighing between 0.3 and 2.15 mg (Valentine 1949), as also verified for the pine forest populations. The investigated deciduous forest populations belong to the pure species Viola riviniana (Neuffer et al. 1999) and exhibit seed weights ranging between 0.3-2.1mg, whereby intermediate seeds are preferably produced. Very light seeds weighing 0.3mg are most probably incapable of germinating. A possible interpretation is that this concerns late-aborted seeds. The direct reason for the abortion of generative organs in general is their competition for resources (Lee 1988). Moreover, the abortion of generative organs, especially of seeds, may be of evolutionary significance. This partly concerns the selective abortion of seeds with incompatible combinations of allels or unfavourable mutations. This opens up the possibility of the differentiated abortion of inferior seeds with an increase in the average quality of seeds, which is likely to play an important role in the old deciduous forest population with habitats isolated over several centuries. Such factors, referred to as “genetic load”, are quoted to be the most important causes of seed abortion in cross-pollinated species (Wiens et al. 1987). Besides favourable microhabitats, the latter would explain the high seedling survival rate in the deciduous forest populations. This underlines the fact that habitat type, population age and other factors have a combined effect, interact with each other, and cannot be treated in isolation. A permanent “overproduction” of flowers or ovules results in high reproduction costs, which is also reflected in the deciduous forest populations over a short ramet life span (high annual turnover).
The demographic population differentiation in the pine forests is also related to genetic differentiation among local Viola populations (Auge et al. 2001). Besides self-pollination, other factors which most probably play a role include short seed dispersal distance, repeated seedling recruitment, genetic drift and founder effects within the population differentiation. The reason for this is that the pine forest populations have most probably colonized their respective sites during the past few centuries (Neuffer et al. 1999). Despite differentiation, the investigations indicate a number of similarities among the populations studied: resource allocation capability and the high reproduction costs arising from this lead to a short ramet life span, which raises the question of an optimum life history strategy. A further explanation of population differentiation is the introgression of a number of Viola reichenbachiana genes into the Viola riviniana populations of the pine forests of the Dübener Heide. Accordingly, the demographic studies are in agreement with the molecular-genetic investigations of Neuffer et al. 1999, under the supposition that introgressive hybridization gives rise to new genotypes adapted to the changed environmental conditions in polluted pine forests. It may be concluded that hybridization is not only an important event in species development but may also be viewed as an important mechanism for the adaptation of populations to a changed environment (cf. also Neuffer et al. 1999). It will be assumed that the Viola populations of the pine forests have “colonized their respective sites during the past few centuries” (in citing Neuffer et al. 1999). But the present results raises the question, of the introgressive hybridization of Viola species described to aerial pollutions occurred not until the last decennia. Viola riviniana and Viola reichenbachiana coexist in the isolated deciduous forest sites within the pine forests of the Dübener Heide, so that from this hybridization and invasion is possible.
The term late-aborted seeds is related to the development stages of the embryo: the seed size is like a ripe seed, but the embryo fills at most 2/3 of the testa. This kind of seeds normally don’t germinate. “Genetic load” is a possible explanation of late-aborted seeds.
Interesting continued literature
Maron, J.L, Vila, M., Bommarco, R., Elmendorf, S., Beardsley, P. (2004): Rapid evolution of an invasive plant. Ecological Monographs, 74(2), pp. 261-280.
Phenotypic Plasticity in Plants
Consequences of Non-Cognitive Behaviour. An international workshop - March 15-19, 1998. Ben Gurion University of the Negev, the Jacob Blaustein Institute for Desert Research.
References
Abbott, R.J. (1992). Plant invasions, interspecific hybridization and the evolution of new plant taxa. Trends in Ecology and Evolution 7, 401-405.
Abrahamson, W.G. & Gagdil, M.D. (1973). Growth form and reproductive effort in golden rods (Solidago, Compositae). American Nature 107: 651-661.
Arnold, M.L. (1997). Natural Hybridization and Evolution. Oxford University Press, New York.
Auge, H. (1991). Experimentelle Untersuchungen zur Populationsökologie von Galium aparine L. (Rubiaceae) – Ökotypendifferenzierung und phänotypische Plastizität pp. 93, Diss. MLU Halle.
Auge, H. (1997). Biologische Invasionen: Das Beispiel Mahonia aquifolium. In: Regeneration und nachhaltige Landnutzung – Konzepte für belastete Regionen (eds. Feldmann, R., Henle, K. & Auge, H.), pp. 124-129, Springer, Berlin.
Auge, H. & Brandl, R. (1997). Seedling recruitment in the invasive clonal shrub Mahonia aquifolium. Oecologia 110, 205-211.
Auge, H., Neuffer, B., Erlinghagen, F., Grupe, R. & Brandl, R. (2001). Demographic and random amplified polymorphic DNA analyses reveal high levels of genetic diversity in a clonal violet. Molecular Ecology 10, 1811-1819.
Beattie, A.J. (1969). The floral biology of three species of Viola. New Phytologist 68, 1187-1201.
Beattie, A.J. & Lyons, N. (1975). Seed dispersal in Viola (Violaceae): adaptions and strategies. American Journal of Botany 62, 714-722.
Berg, H. & Redbo-Torstensson, P. (1999). Offspring performance in three cleistogamous Viola species. Plant Ecology 145, 49-58.
Bierzychudek, P. (1982). Life histories and demography of shade-tolerant temperate forest herbs: a review. New Phytologist 90, 757-776.
Cody, M.L. (1966). A general theory of clutch size. Evolution 20, 174-184.
Cooke, R.E. (1985). Growth and Development in Clonal Plant Populations. In: Jackson, J.B.C., Buss, L.W. & Cooke, R.E. Population Biology and Evolution of Clonal Organisms.pp. 65-83,Yale University Press, New Harenn and London.
Daehler, C.C. & Strong, D.J. (1997). Hybridization between introduced smooth cordgrass (Spartina alterniflora, Poaceae) and native California cordgrass (Spartina foliosa) in San Francisco Bay, California, USA. American Journal of Botany 84, 607-611.
Eriksson, O. (1989). Seedling dynamics and life histories in clonal plants. Oikos 55, 231-238.
Eriksson, O. (1993). Dynamics of genets in clonal plants. Trends in Ecology and Evolution 8, 313-316.
Eriksson, O. & Ehrlen, J. (1992). Seed and microsite limitation of recruitment in plant populations. Oecologia 91, 360-364.
Erlinghagen, F. (1995). Populationsökologische Untersuchungen an Viola riviniana RCHB. in unterschiedlich stark geschädigten Kiefernforsten der Dübener Heide (Sachsen-Anhalt). Kieler Notizen 23, 16-19.
Erlinghagen, F. (1998). Populationsökologische Untersuchungen an Viola riviniana RCHB. in immissionsgeschädigten Kiefernforsten der Dübener Heide (Sachsen-Anhalt). Unpublished manuscript, pp. 90.
Erlinghagen, F. (2004). Comparative investigations of the spatial and temporal population dynamics and morphology of the clonal hybrid Viola riviniana RCHB. x Viola reichenbachiana JORDAN ex BOR. in polluted pine forests of the Dübener Heide (Saxony-Anhalt). 17th workshop of the GfÖ working group “Population biology of plants” in Regensburg, Germany.
Fitter, A.H. & Setters N.L. (1988). Vegetative and reproductive allocation of phosphorus and potassium in relation to biomass in six species of Viola. Journal of Ecology 76, 617-636.
Grime, J.P. (1979) Plant strategies and vegetation process, pp. 217. Cichester: Wiley & Sons.
Grime, J.P., Hodgson, J.G. & Hunt, R. (1988). Comparative Plant Ecology. London: Unwin Hyman.
Greig-Smith, P. (1983). Quantitative Plant Ecology. Berkeley: University of California Press.
Große, E. (1985). Beiträge zur Geschichte der Wälder des Stadtkreises Halle und des nördlichen Saalkreise. Hercynia N.F. 22 , 37-52. Leipzig.
Haig, D. & Westoboy, M. (1988). Inclusive fitness, seed resources, and maternal care. In: Lovett-Doust, J. & Lovett-Doust, L. (eds.): Plant Reproductive Ecology, pp. 60-79. New York: Oxford Univ. Press.
Harper, J.L. & Ogden, J. (1970). The reproductive strategy of higher plants. I. The concept of strategy with special reference to Senecio vulgaris. Journal of Ecology 58, 681-698.
Inghe, O. & Tamm, C. (1985). Survival and flowering of perennial herbs. IV. The behaviour of Hepatica nobilis and Sanicula europaea on permanent plots during 1943 and 1981. Oikos 45, 400-420.
Klimes, L., Klimesova, J., Hendriks, R. & van Groenendael, J. (1997). Clonal plant architecture: a comparative analysis form and function. In: The Ecology an Evolution of Clonal Plants (eds. Dekroon, H. & van Groenendael, J.), pp. 1-29. Leiden: Backhuys Publishers.
Krebs, C.J. (1989). Ecological Methodology, pp. 77-78. Univ. of British Columbia, Harper Collins-Publishers.
Lovett-Doust, L. (1981). Population dynamics and local specialization in a clonal perennial (Ranunculus repens). I. The dynamics of ramets in contrasting habits. Journal of Ecology 69, 743-755.
Lee, T.D. (1988). Patterns of seed and fruit production. In: Lovett-Doust, J. & Lovett-Doust, L. (eds.): Plant Reproduction Ecology – Patterns and Strategies, pp. 179-202. New York: Oxford Univ.Press.
Luken, J.O. (1987). Interactions between seed production and vegetative growth in the staghom sumac, Rhus typhina L. Bull. Torrey Bot. Club 114, 247-251.
Meusel, H. & Kästner, A. (1974). Zur Wuchsform einiger Veilchenarten. Phyton (Austria) 16, 127-135.
Meusel, H. , Jäger, E. , Rauschert, S. & Weinert, E. (1978). Vergleichende Chorologie der zentraleuropäischen Flora, Vol.II: Text. Jena: Gustav Fischer.
Müller-Schneider, P. (1986). Verbreitungsbiologie der Blütenpflanzen Graubündens. Veröf. Geobot.Inst.ETH 85, Zürich.
Neuffer, B., Auge, H., Mesch, H., Amarell, U. & Brandl, R. (1999). Spread of violets in polluted pine forests: morphological and molecular evidence for the ecological importance of interspecific hybridization. Molecular Ecology 8, 365-377.
Neumeister, H., Franke, C., Nagel, C., Peklo, G., Zierath, R. & Peklo, P. (1991). Immissionsbedingte Stoffbeiträge aus der Luft als geomorphologischer Faktor. – Geodynamik XII, 1-40.
Newell, S.J., Solbrig, O.T & Kincaid, D.T. (1981). Studies on the population biology of the genus Viola. III. The demography of Viola blanda and Viola pallens. Journal of Ecology 69, 997-1016.
Oberdorfer, E. (1990). Pflanzensoziologische Exkursionsflora. 6.Aufl. Stuttgart: Eugen Ulmer.
Rieseberg, L.H. (1995). The role of hybridization in evolution: old wine in new skins. American Journal of Botany 82, 944-953.
Rothmaler, W. (1996). Exkursionsflora von Deutschland, Vol.2, Gefäßpflanzen: Grundband. 16.Aufl. Jena: Gustav Fischer.
Scheiner, S.M. (1993). Genetics and evolution of phenotypic plasticity. Annual Review of Ecology Syst. 24, 35-68.
Silvertown, J., Franco, M., Pisanty, I. & Mendoza, A. (1993). Comparative plant demography - relative importance of life-cycle components to the finite rate of increase in woody and herbaceous perennials. Journal of Ecology 81, 465-476.
Solbrig, O.T. (1981). Studies on the plant size on fitness in Viola sororia. Evolution 66, 123-134.
Solbrig, O.T. & Simpson, B.B. (1974). Components of regulation of a population of dandelions in Michigan. Journal of Ecology 62, 473-486.
Stace, C.A. (1975) Hybridization and the Flora of the British Isles. London: Academic Press.
Stearns, S.C. (1976). Life-history tactics: a review of the ideas. Quart.Rev.Biol. 51, 3-47.
Turnbull, L.A., Crawley, M.J. & Rees, M. (2000). Are plant populations seed-limited ? A review of seed sowing experiments. Oikos 88, 225-238.
Urbanska, K.M. (1992). Populationsbiologie der Pflanzen. Stuttgart: Gustav Fischer.
Valentine, D.H. (1941). Variation in Viola riviniana RCHB. New Phytologist 40, 189-209.
Valentine, D.H. (1949). The experimental taxonomy of two species of Viola. New Phytologist 49, 194-212.
Watkinson, A.R. & Powell, J.C. (1993). Seedling recruitment and the maintenance of clonal diversity in plant populations – a computer simulation of Ranunculus repens. Journal of Ecology 81, 707-717.
Wiegand, S. & Amarell, U. (1995). Ecological studies in a pollution gradient: Do plant and animal communities respond differently ? Archiv für Landschaftsforschung und Naturschutz 33, 271-286.
Wiens, D., Calvin, C.L., Wilson, C., Davern, C.A., Frank, D. & Seavey, S.R. (1987). Reproductive success, spontaneous embryo abortion, and genetic load in flowering plants. Oecologia 71, 501-509.
Friederike Erlinghagen works on population ecology, community ecology and on plant-insect interactions (e.g. honeydew producer, Hymenoptera Apoidea)